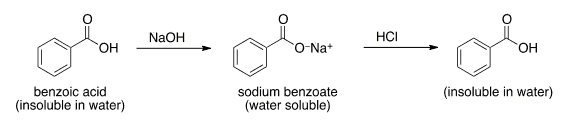
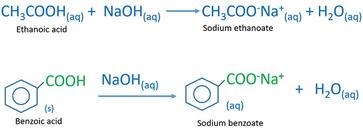
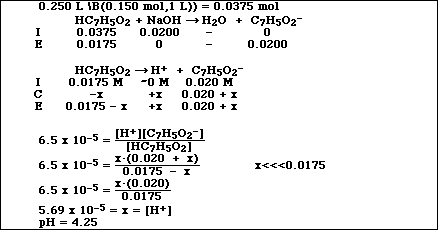SOLVED: A solution of benzoic acid (HC,HsOz) is titrated using sodium hydroxide: The Ka of benzoic acid is 6.4x 101. Write the balanced equation and the net ionic equation. 2.100.0 mL of

1. Draw a balanced chemical equation for the reaction that would occur between benzoic acid and aqueous sodium hydroxide. 2. Draw a balanced chemical equation for the reaction that would occur between

Explain the results for the tube in which 1.0 m naoh was added to benzoic acid. write an equation for this, - Brainly.com
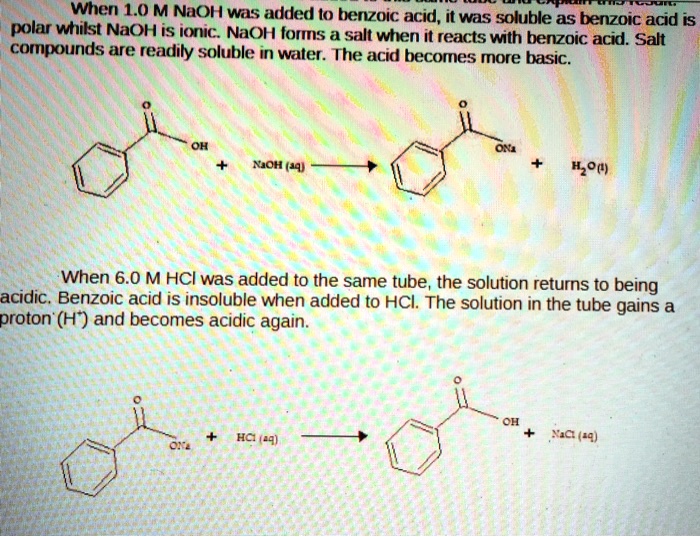
SOLVED: When 1.0 M NaOH was added (0 benzoic acid, it was soluble as benzoic acid is polar wtitst NaOH is ioric; NaOH forms salt when i reacts with berizoic aczo Sad

Titration curves of benzoic acid for different analyte concentrations... | Download Scientific Diagram

When a solution of benzoic acid was titrated with `NaOH` the `pH` of the solution when half - YouTube
![When a solution of benzoic acid was titrated with NaOH the pH of the solution when half the acid neutralized was 4.3 . Dissociation constant of the acid is [Use log 2 = 0.3] . When a solution of benzoic acid was titrated with NaOH the pH of the solution when half the acid neutralized was 4.3 . Dissociation constant of the acid is [Use log 2 = 0.3] .](https://haygot.s3.amazonaws.com/questions/1835171_829774_ans_40c76abc129644cfaff5cbb818338569.png)
When a solution of benzoic acid was titrated with NaOH the pH of the solution when half the acid neutralized was 4.3 . Dissociation constant of the acid is [Use log 2 = 0.3] .

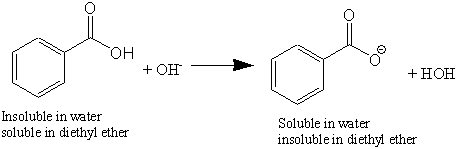
OneClass: Benzoic acid is soluble in diethyl ether but not water, however, benzoic acid is extracted ...

Experiment 6 Extraction - Separation of Benzoic Acid and Phenanthrene - Experiment 6: Extraction - - Studocu

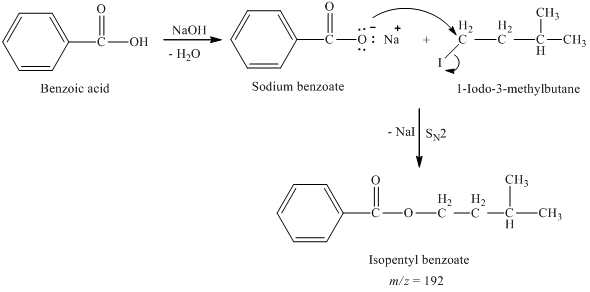
Write the mechanism for the reaction of either benzoic acid or acetic acid with NaOH. Be sure to include all major structures and resonance forms. | Homework.Study.com

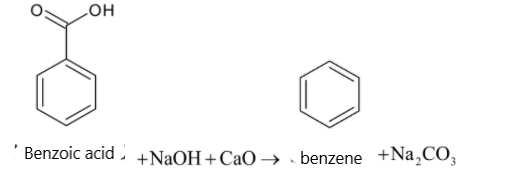
For the given reaction :2 phenylacetic acid + NaOH CaO , ⟶ ProductThe product is:A. Benzoic acidB. TolueneC. PhenolD. Benzene

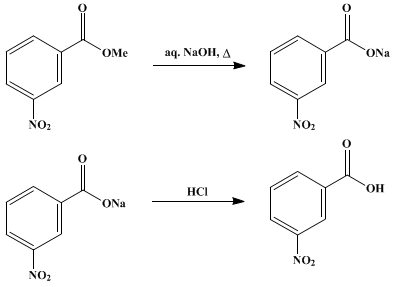
Write a balanced equation for the reaction of benzoic acid with hydroxide ion. Why is it necessary to extract the ether layer with sodium hydroxide? | Homework.Study.com

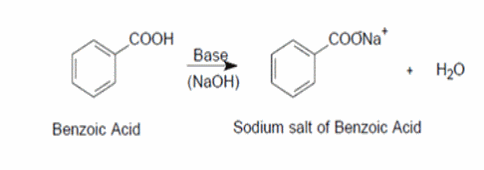
Draw the products of benzoic acid reacting with sodium hydroxide. Draw the products of the pyridine reacting with hydrochloric acid. Use the "+/-" button to add the charge (and H atom).
✓ Solved: Explain the results for the tube in which 1.0 M NaOH was added to benzoic acid. Write an equation...