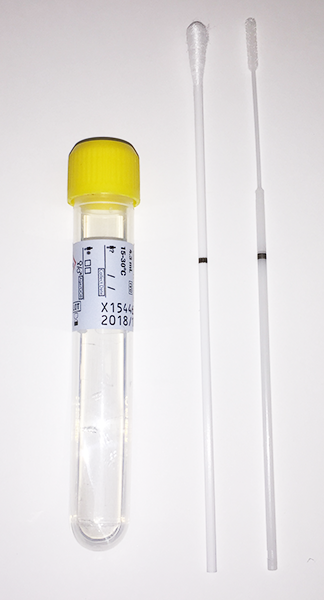
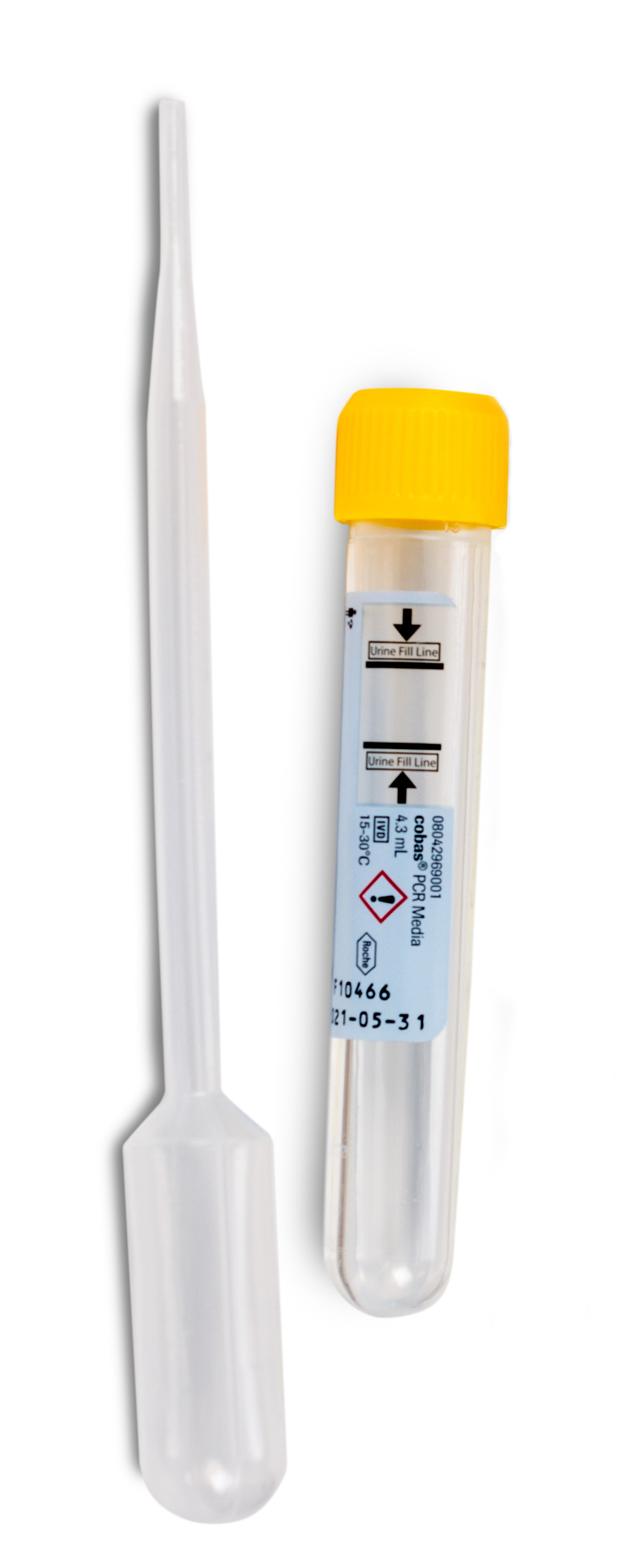
Molecular Testing for Sexually Transmitted Infections: Phase 3 Transition to Roche Method and Collection Devices

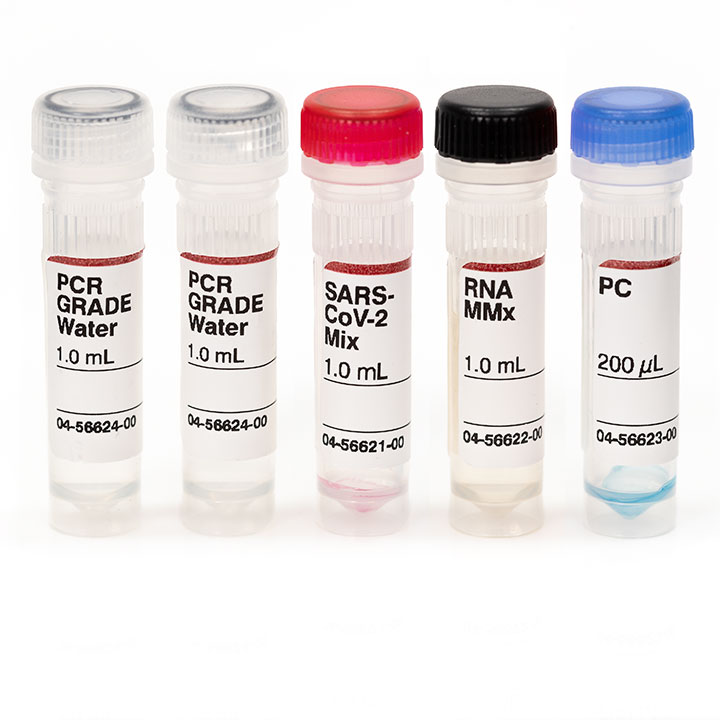
First-in-Class Study Validates Alternate Transport Media and Broader Storage Conditions for SARS-CoV-2 (COVID-19) Testing on RT-PCR Platforms | COPAN Diagnostics Inc.

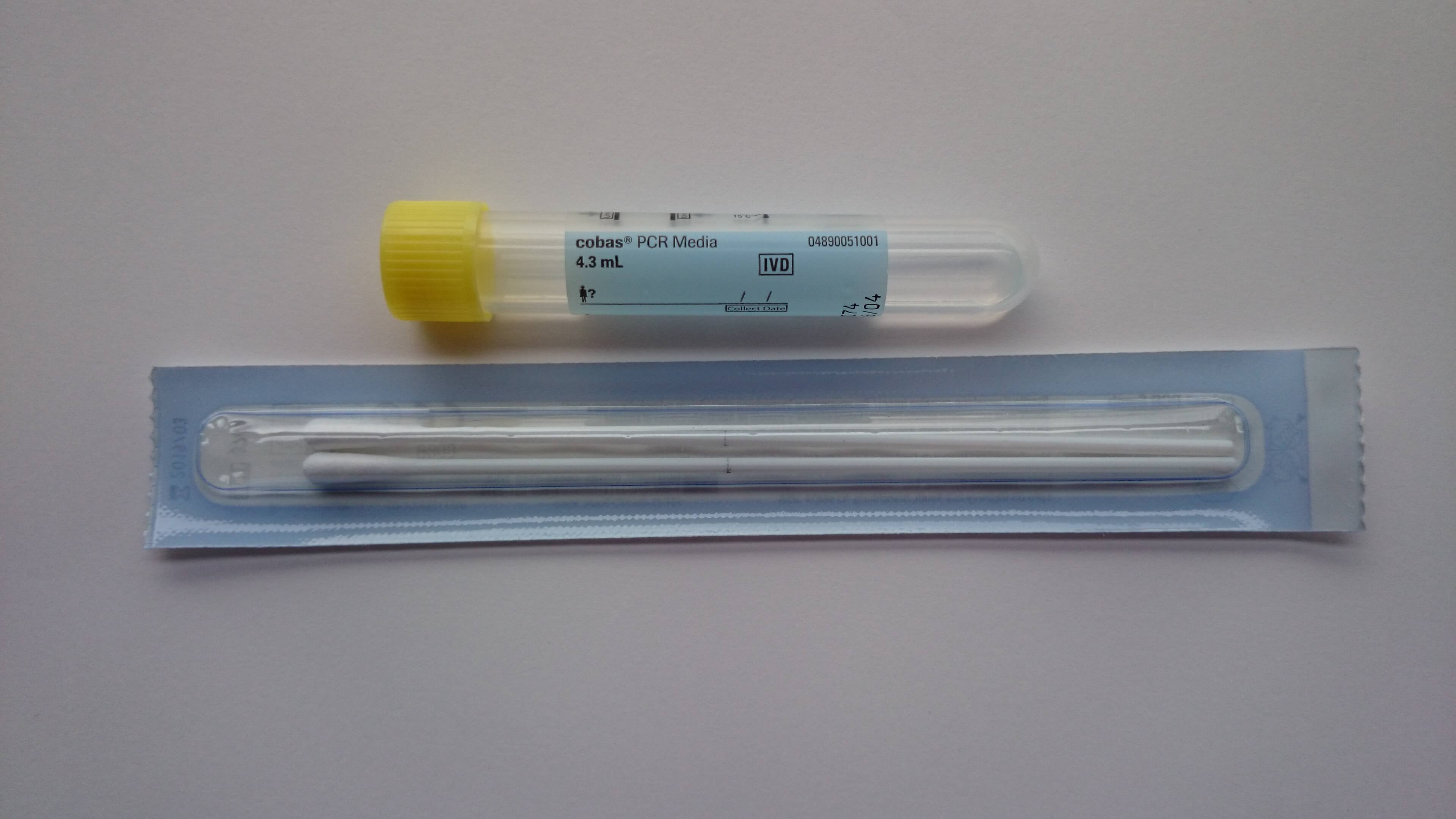
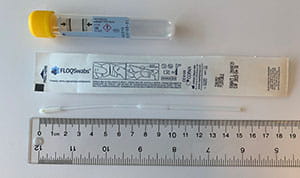
Chlamydia trachomatis/Neisseria gonorrhoeae (CT/NG) – Nucleic Acid Amplification Testing (NAAT) | Public Health Ontario