Which should be stronger acid, HOCN, or HCN? Explain briefly In HOCN, the H+ ion is attached to th - YouTube

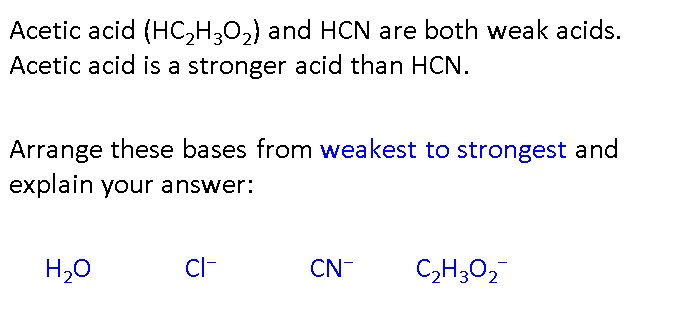
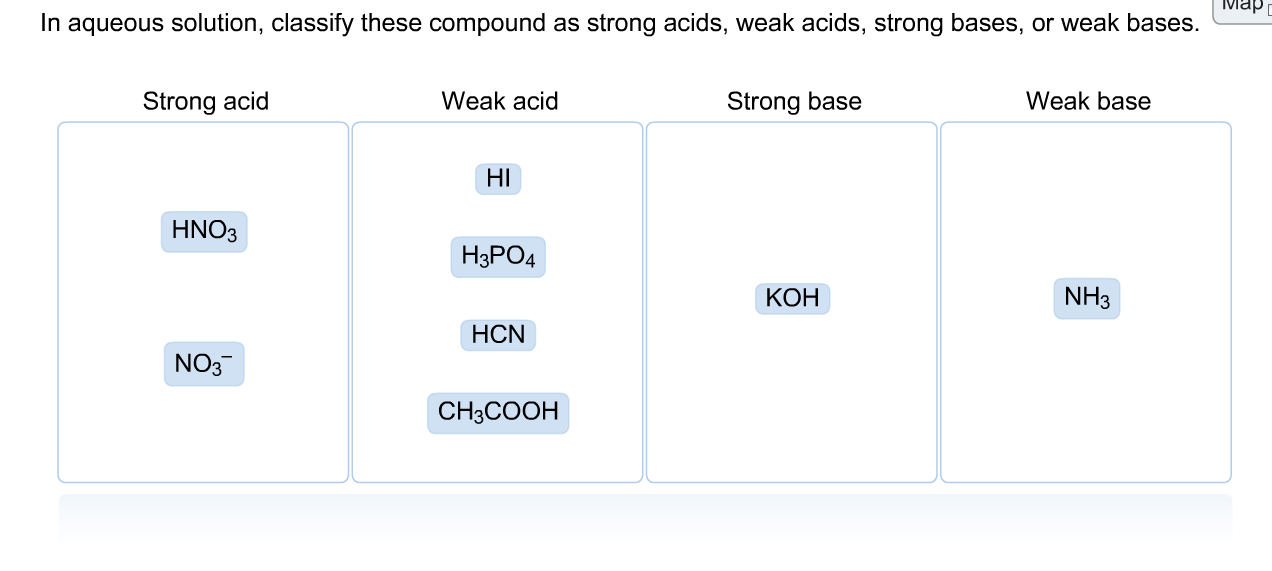
Identify the stronger acid in each pair: Part A: NH^+4 or H3O^+ Part B: H2SO4 or HCN Part C: H2O or H2CO3 | Homework.Study.com
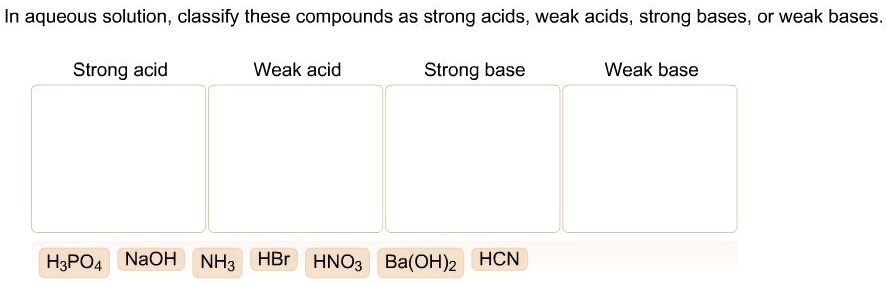
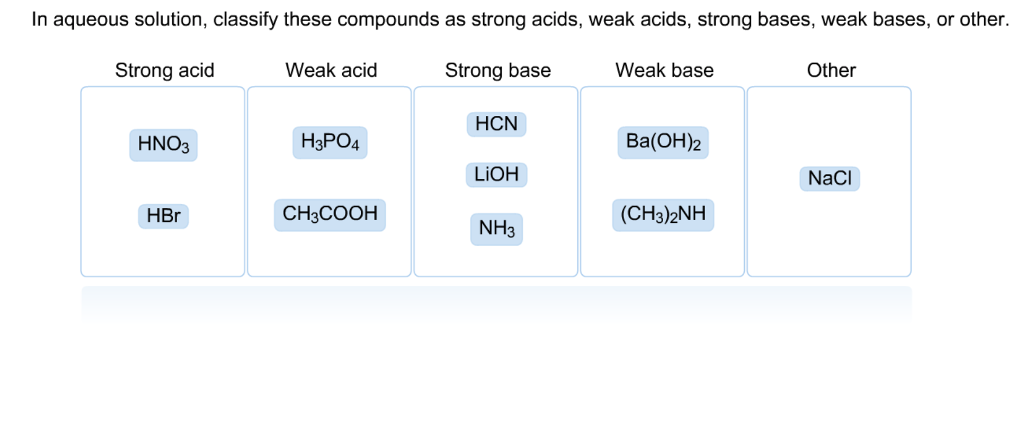
SOLVED: In aqueous solution, classify these compounds as strong acids, weak acids, strong bases or weak bases Strong acid Weak acid Strong base Weak base HaPO4 NaOH NH3 HBr HNO3 Ba(OH)z HCN

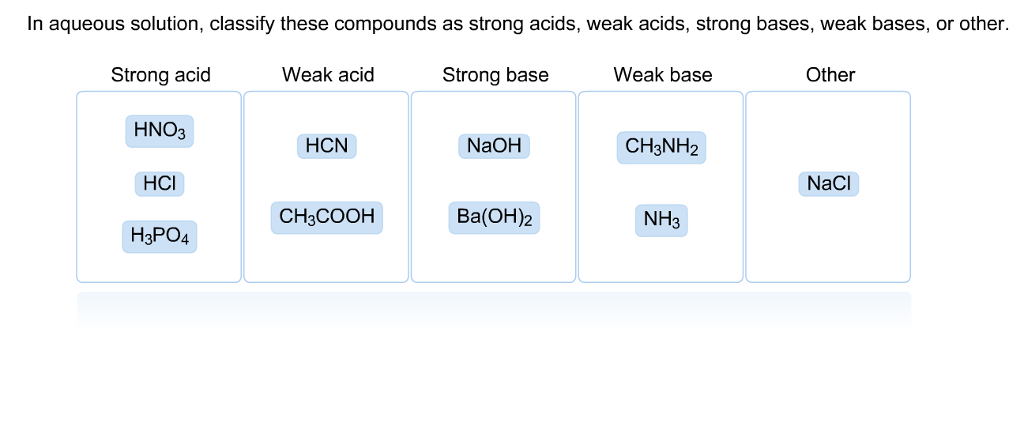
SOLVED: In aqueous solution, classify these compounds a5 strong acids, weak acids, strong bases, Or weak bases Strong acid Weak acid Strong base Weak base H;POA HBr HCN NaOH CH,COOH HNO; Br "

HCN is a weak acid ( Ka = 6.2 × 10^-10 ) ,NH4OH is a weak base ( Kb = 1.8 × 10^-5 ) . A 1.00 M solution of NH4CN would be:

Hydrogen cyanide (HCN)- Lewis acid Structure, Molecular mass, Physical and Chemical Properties, Uses with FAQs of Hydrogen Cyanide.

HCN is a weak acid ( Ka = 6.2 × 10^-10 ) ,NH4OH is a weak base ( Kb = 1.8 × 10^-5 ) . A 1.00 M solution of NH4CN would be:

The heat of neutralization of a strong base and a strong acid is 13.7 kcal. The heat released when 0.6 mole HCl solution is added to 0.25 mole of NaOH is: