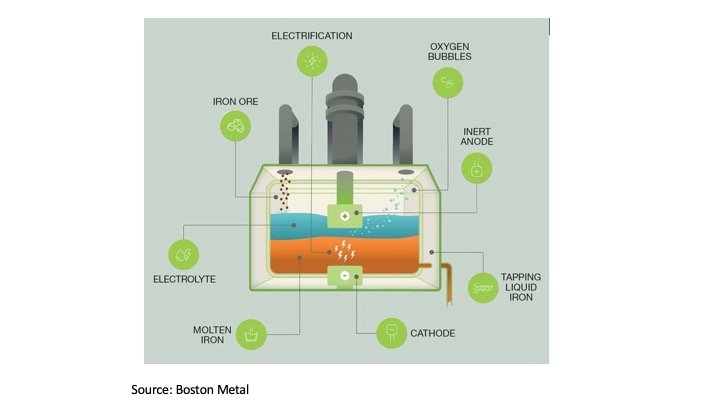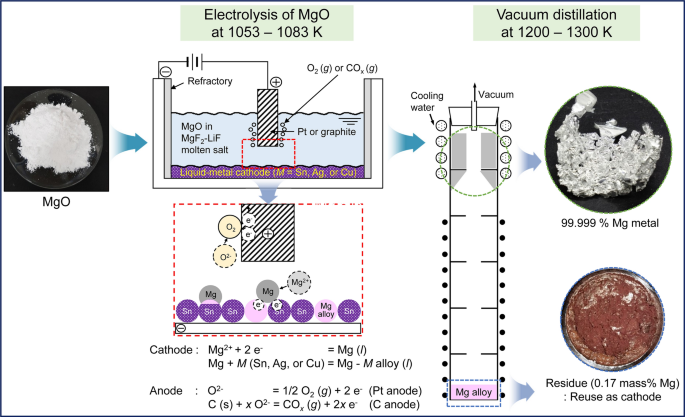
Molten Salt Electrolysis of Magnesium Oxide Using a Liquid–Metal Cathode for the Production of Magnesium Metal | SpringerLink

Electrochemical Reduction of Carbon Dioxide and Iron Oxide in Molten Salts to Fe/Fe3C Modified Carbon for Electrocatalytic Oxygen Evolution - Liang - 2021 - Angewandte Chemie International Edition - Wiley Online Library
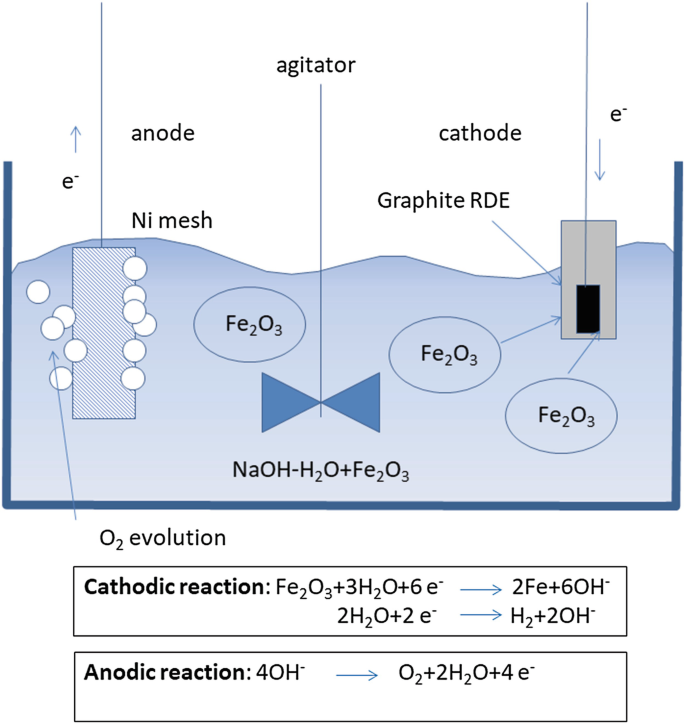
Electrolysis of Iron Ores: Most Efficient Technologies for Greenhouse Emissions Abatement | SpringerLink
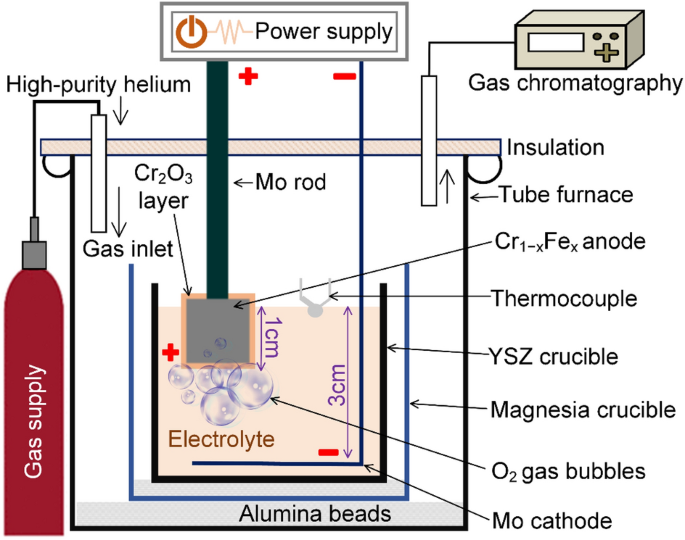
Oxidation and electrical properties of chromium–iron alloys in a corrosive molten electrolyte environment | Scientific Reports
Why can the oxides of Na, Mg, and Al undergo electrolysis, but oxide of S, P, and Cl do not? - Quora

Ultra‐High Temperature Molten Oxide Electrochemistry - Wang - 2022 - Angewandte Chemie International Edition - Wiley Online Library
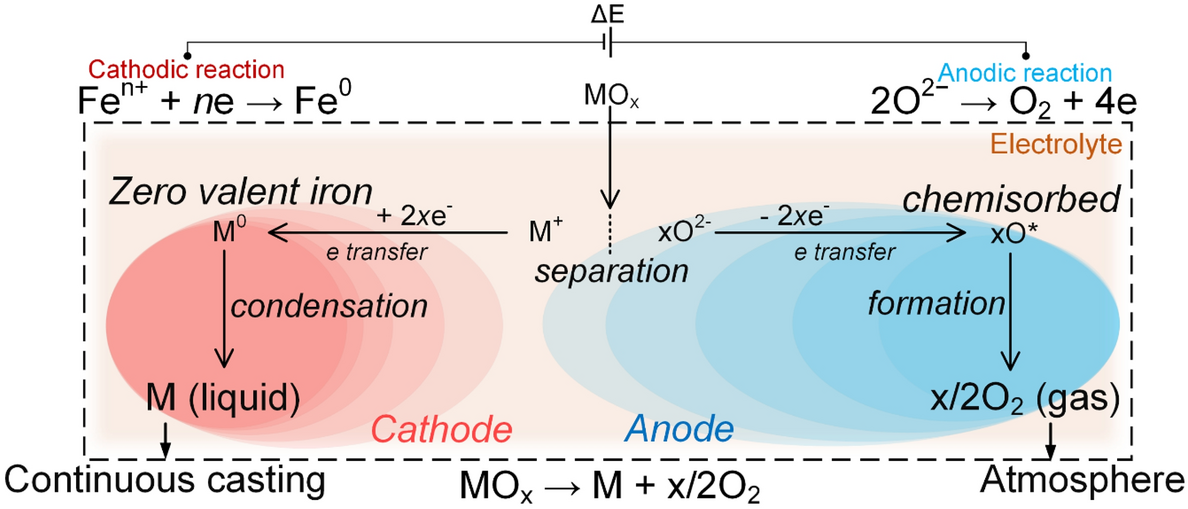
Oxidation and electrical properties of chromium–iron alloys in a corrosive molten electrolyte environment | Scientific Reports
a) Possible pathways to reduce iron (Fe) from iron oxide (Fe 2 O 3 or... | Download Scientific Diagram


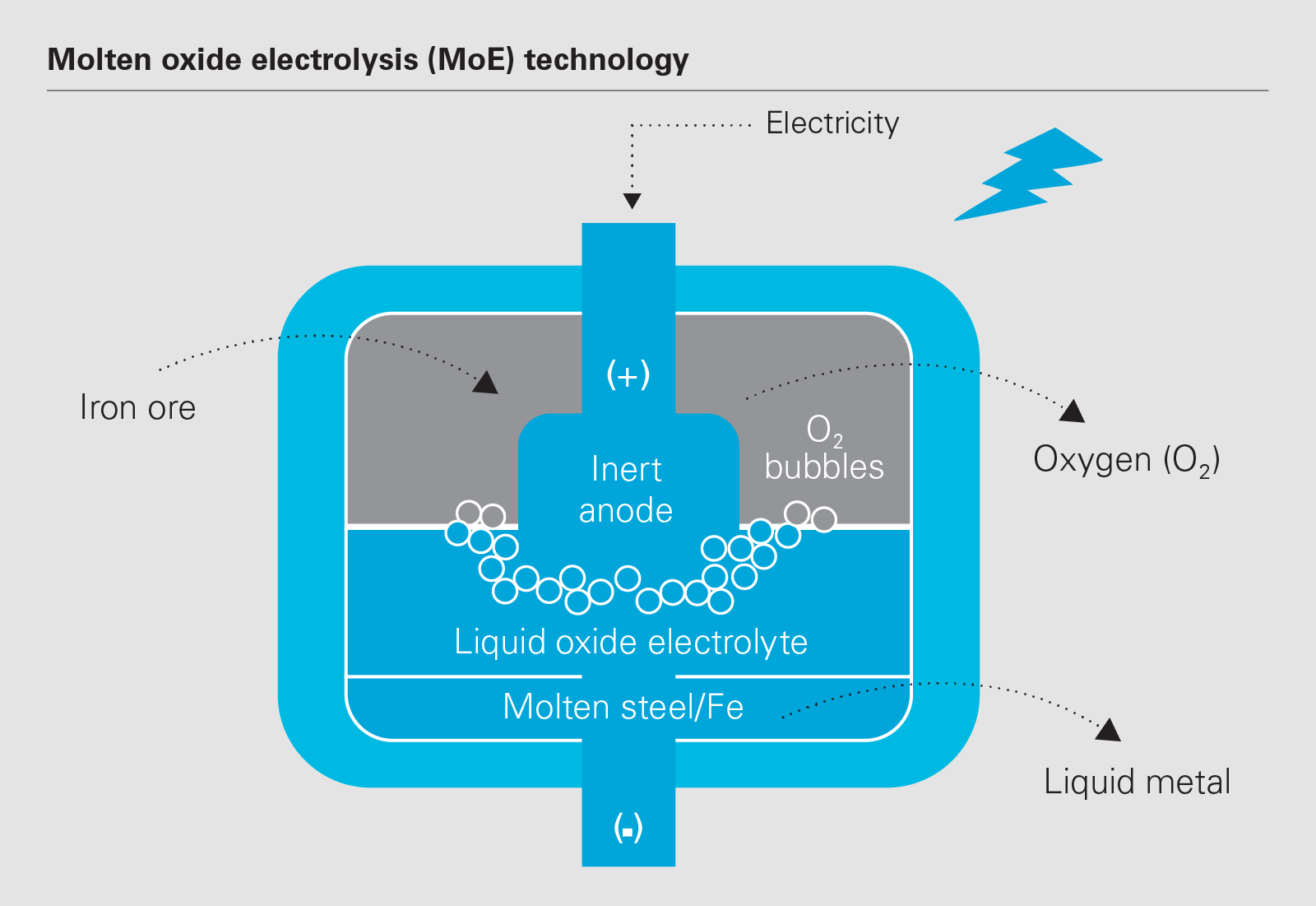
:max_bytes(150000):strip_icc()/TappingBostonMetalsMOEcell-7f0a3a5b63d84be0982289e865788721.jpg)
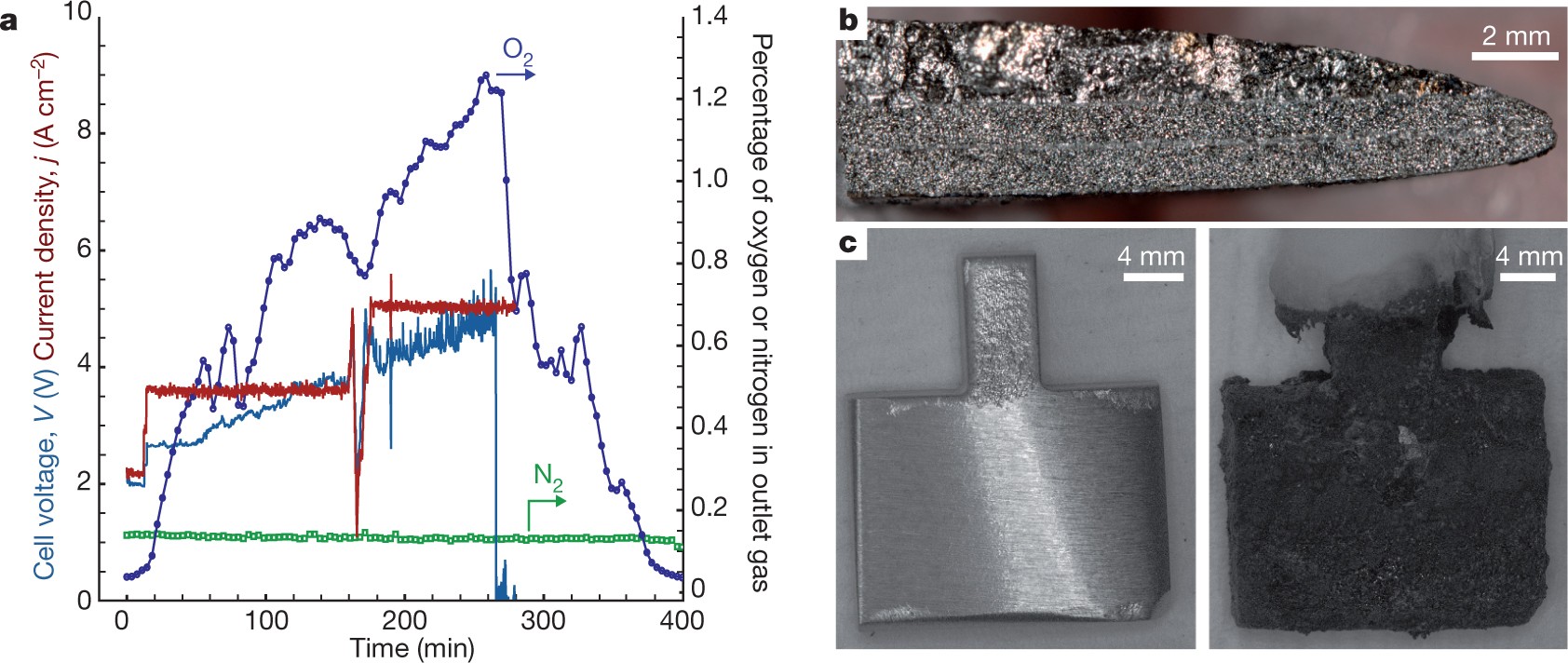
.jpg)