Write balanced chemical equation for the following : 1. Reaction of nitric acid with sodium bicarbonate. - Sarthaks eConnect | Largest Online Education Community
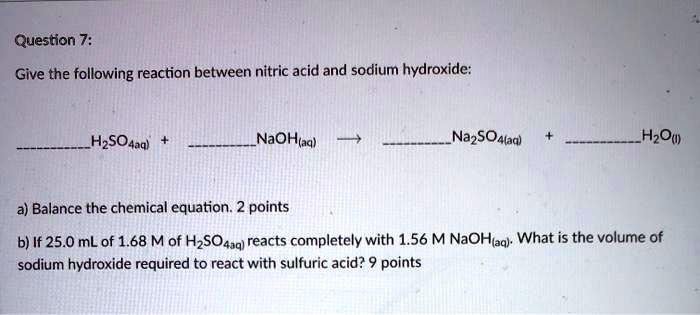
SOLVED: Question 7: Give the following reaction between nitric acid and sodium hydroxide: HzSOAaal NaOHtaq) NazSOAlaa) HzOu) a) Balance the chemical equation. 2 points b) If 25.0 mL of 1.68 Mof HzSO4uq)

SOLVED: An aqueous solution of nitric acid (HNO3) is standardized by titration with a 0.311 M solution of sodium hydroxide (NaOH): HNOz + NaOH 77 NaNOz + H2O If14.4 mL of base

Ionic equations A chemical equation shows the number of atoms and molecules of the reactants and products. Also shows physical state of reactants and products. - ppt download

Write balanced chemical equations for the reaction between calcium bicarbonate and dilute hydrochloric acid.

SOLVED: Refer to the neutralization of nitric acid by sodium hydroxide discussed in the Introduction. Calculate the heat of reaction (in kJ) when 40.7 mL of 1.2 M nitric acid reacts with

HNO3 + NaOH = NaNO3 + H20 Balanced Equation||Nitric Acid + Sodium Hydroxide Balanced Equation - YouTube

The pH titration curves of nitric acid (C 0 a = 0.0002 mol l −1 ) being... | Download Scientific Diagram

Which of the following statements are correct ? (i) Boron reacts with concentrated HNO3 to form nitric oxide and boric acid (ii) Boron reacts with fused NaOH to form H2O2 and boric

a) What happens when ethanoic acid reacts with sodium hydroxide ? Write equation of he reaction - YouTube

Experimental results from the titration experiments of nitric acid and... | Download Scientific Diagram















