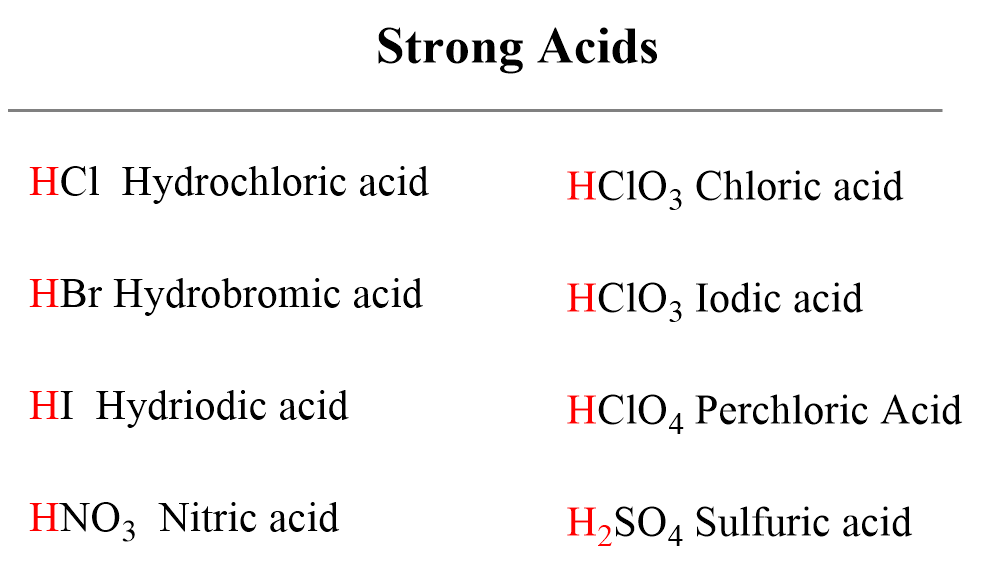
What is the pH of a 0.40 M solution of sodium nitrite, NaNO2? The pKa for nitrous acid (HNO2) is 3.35 - Quora

The ionization constant of nitrous acid is 4.5 × 10^-4 . Calculate the pH of 0.04 M sodium nitrite solution and also its degree of hydrolysis.

Superoxide and Nitrous Acid Production from Nitrate Photolysis Is Enhanced by Dissolved Aliphatic Organic Matter | Environmental Science & Technology Letters

DFT and AIM Study of the Protonation of Nitrous Acid and the pKa of Nitrous Acidium Ion | The Journal of Physical Chemistry A
![PDF] UV-visible spectrum of nitrous acid in solution: pKa determination and analytical applications | Semantic Scholar PDF] UV-visible spectrum of nitrous acid in solution: pKa determination and analytical applications | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7d7622ce83d25c305576ab00e3af3523a05148a3/3-Figure1-1.png)
PDF] UV-visible spectrum of nitrous acid in solution: pKa determination and analytical applications | Semantic Scholar

Microscale pH variations during drying of soils and desert biocrusts affect HONO and NH3 emissions | Nature Communications

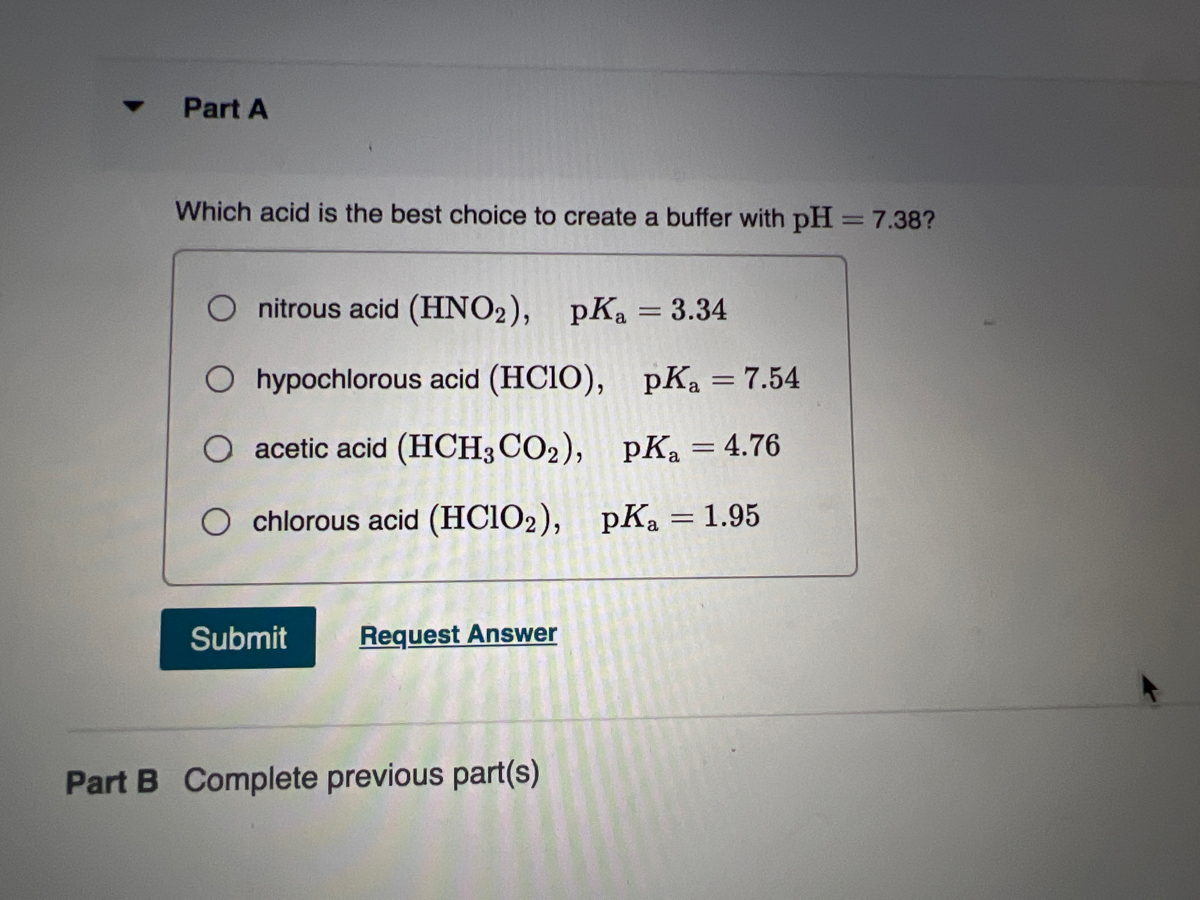
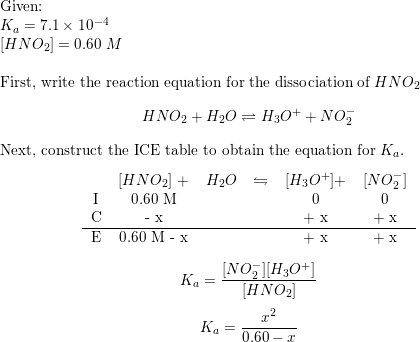

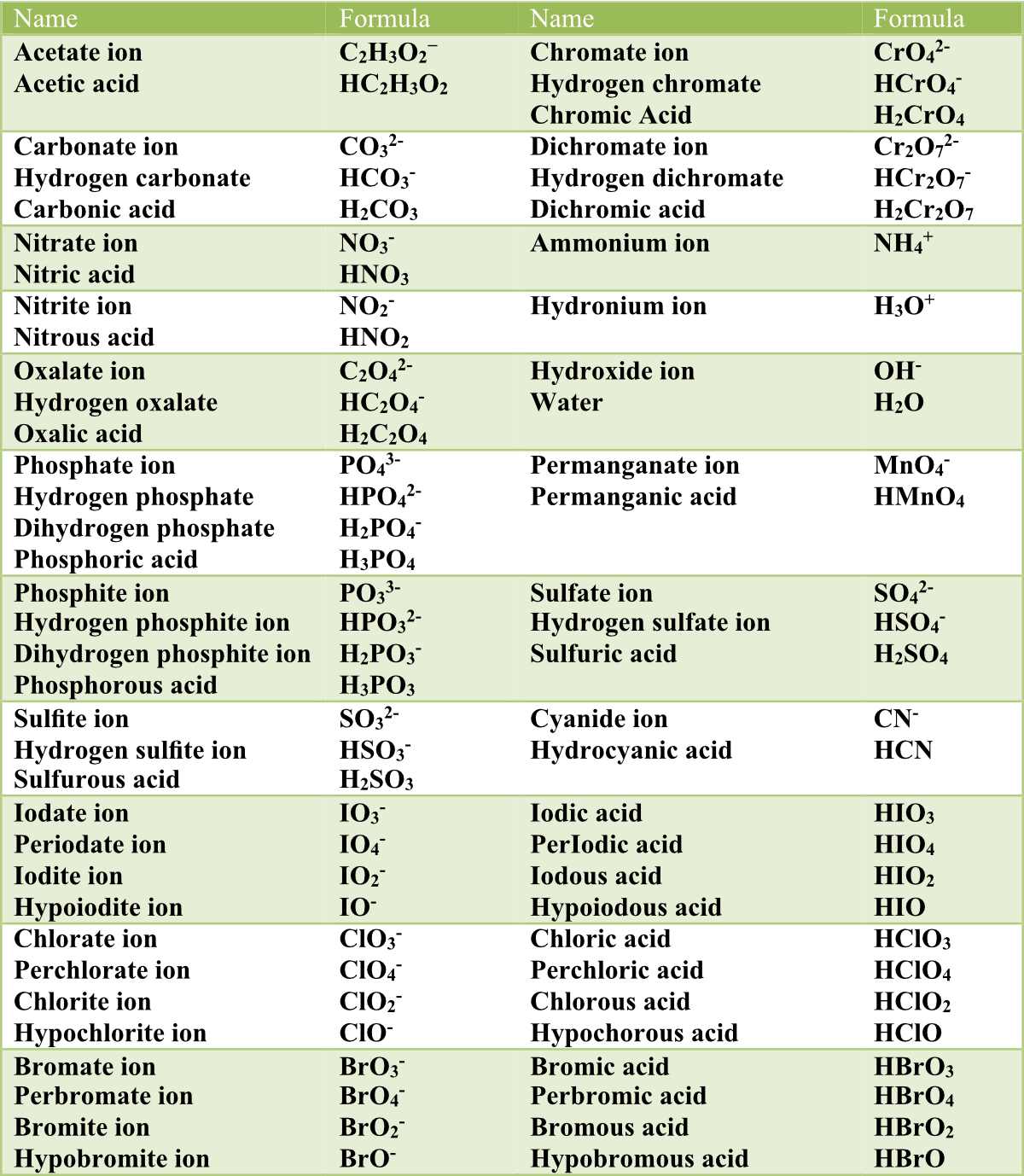



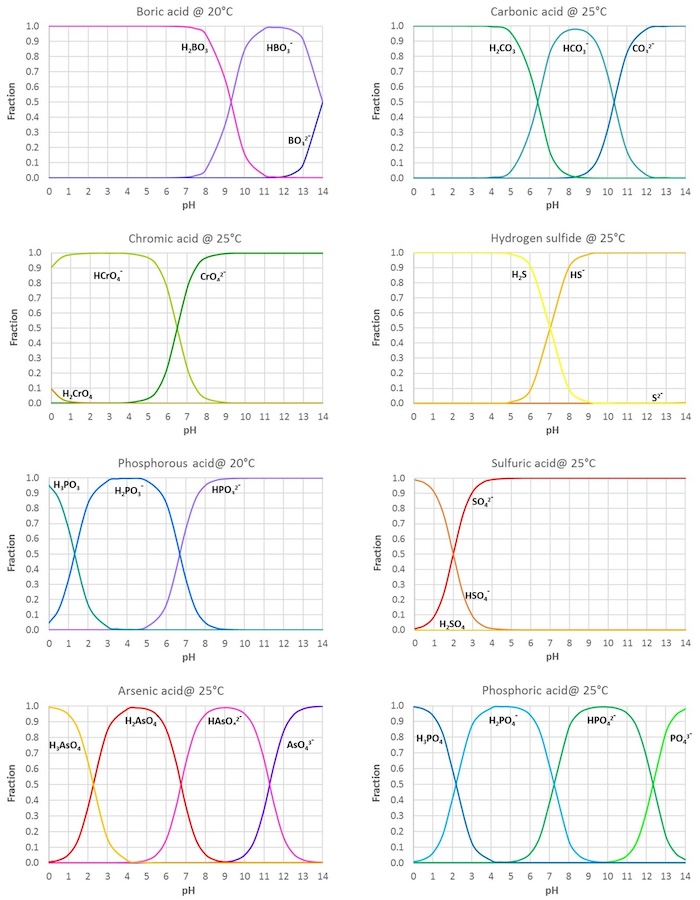

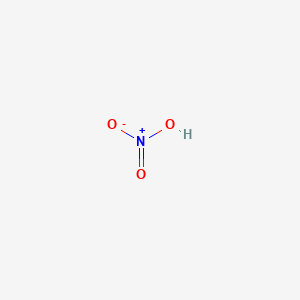


![ANSWERED] Use the pka table to select a base to depr... - Organic Chemistry ANSWERED] Use the pka table to select a base to depr... - Organic Chemistry](https://media.kunduz.com/media/sug-question/raw/80475089-1659894957.2799208.jpeg)