Iron doped Zinc Oxide (Fe 0.2 Zn 0.8 O) by sol-gel method and its Characterization. K. Peddanna By M.Sc. Nano Science Dept. of Physics OSMANIA UNIVERSITY. - ppt download

SOLVED: Zinc oxide adopts a face-centered cubic arrangement. Both the zinc ions and the oxide ions adopt the face-centered cubic arrangement, with the unit cell defined by the cations overlapping the unit

Supported and un-supported zinc and chromium oxide catalysts for lower temperature CO oxidation: A review - ScienceDirect

1: The crystal structures (left to right) zinc blende (GaSb), wurtzite... | Download Scientific Diagram

Large Scale Laser Crystallization of Solution-based Alumina-doped Zinc Oxide (AZO) Nanoinks for Highly Transparent Conductive Electrode | Scientific Reports

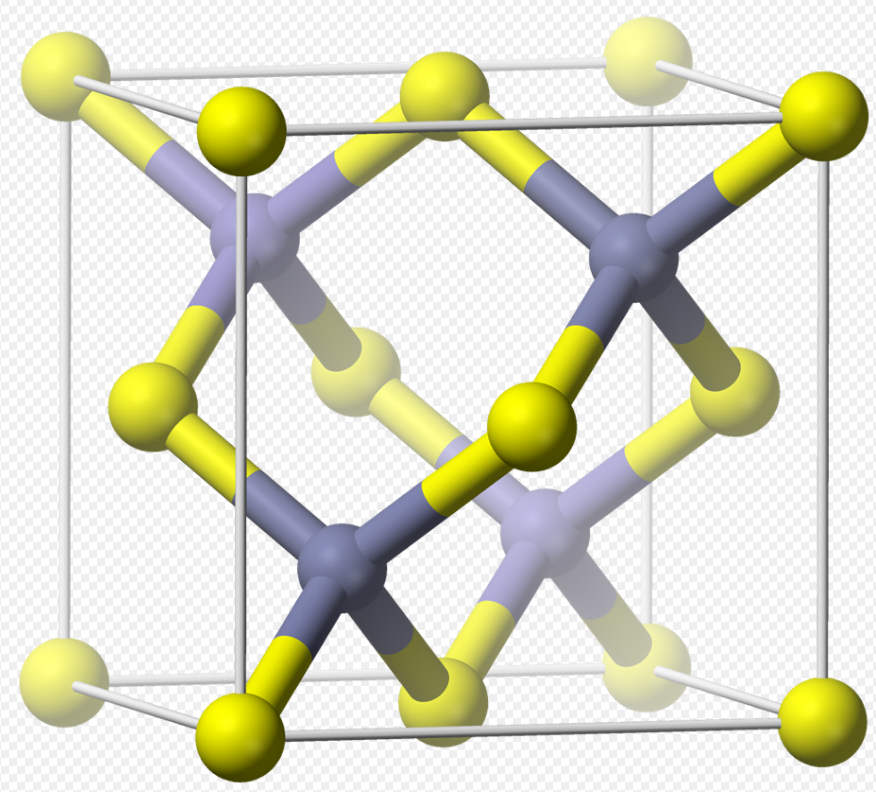
Zinc-blende structure with cubic and hexagonal unit cells. (Reprinted... | Download Scientific Diagram
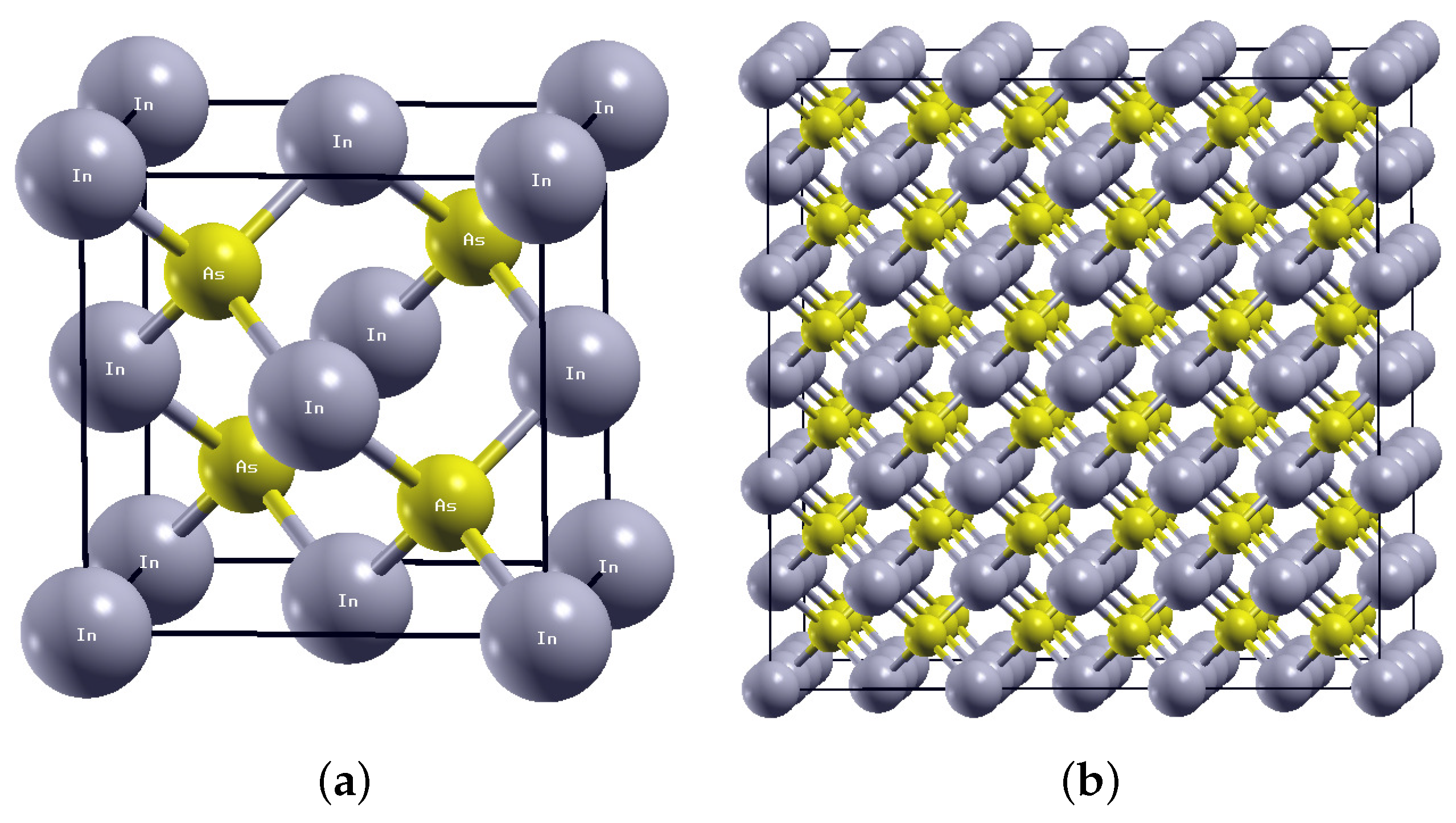
Crystals | Free Full-Text | First-Principles Assessment of the Structure and Stability of 15 Intrinsic Point Defects in Zinc-Blende Indium Arsenide

AgI crystallizes in the ccp, zinc blende structure. Assuming that I^ ions occupy the lattice points, the fraction of tetrahedral voids occupied by Ag^ + ions is :
![Crystal structure of ZnO. a. Zinc Blende, b. Rocksalt c. Wurtzite [5]. | Download Scientific Diagram Crystal structure of ZnO. a. Zinc Blende, b. Rocksalt c. Wurtzite [5]. | Download Scientific Diagram](https://www.researchgate.net/publication/354529911/figure/fig1/AS:1076567929290757@1633685204496/Crystal-structure-of-ZnO-a-Zinc-Blende-b-Rocksalt-c-Wurtzite-5.png)
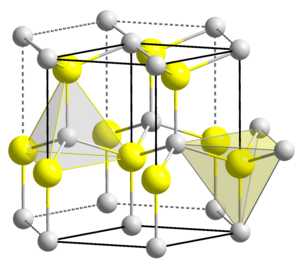
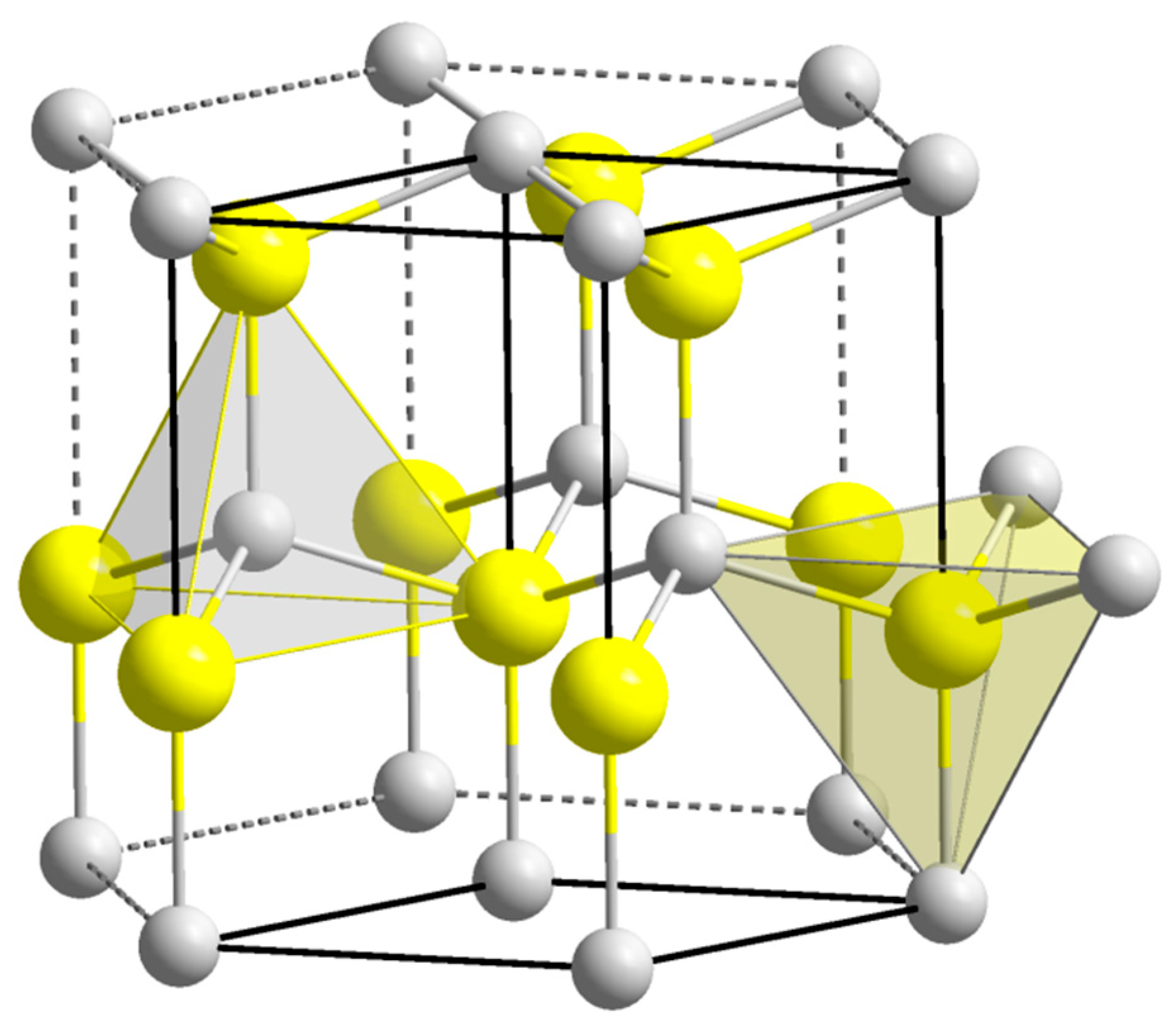
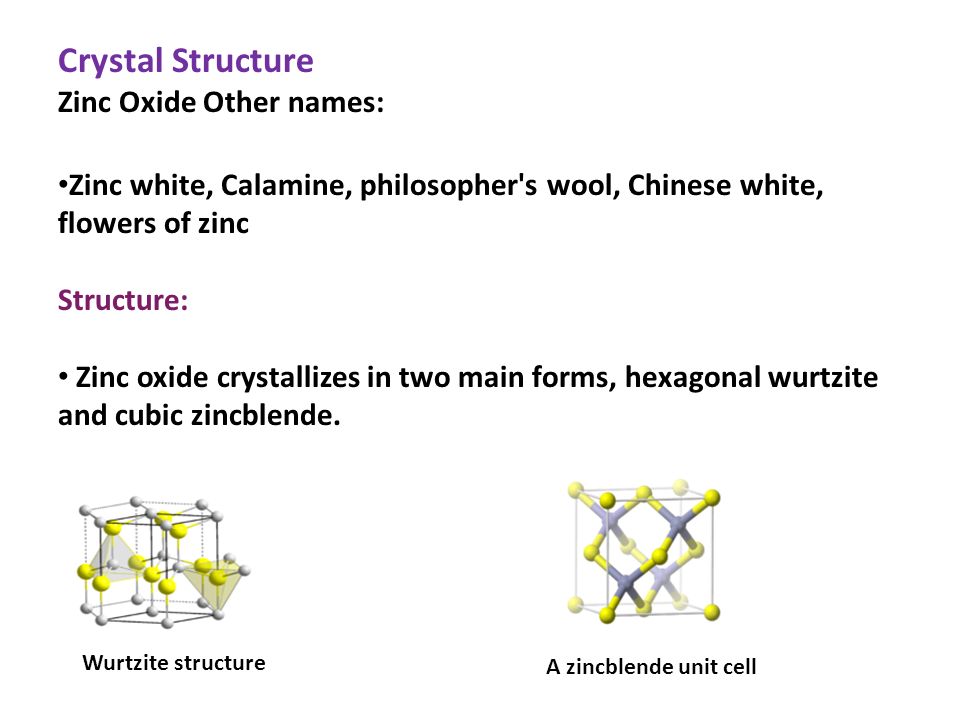
Crystal structure of ZnO. a. Zinc Blende, b. Rocksalt c. Wurtzite [5]. | Download Scientific Diagram

a) Wurtzite crystal structure and (b) zinc blende crystal structure of... | Download Scientific Diagram

In zinc blende structure anions are arranged in ccp and cations are present in the tetrachedral voids and only half the tetrahedral voids are occupied, if 'a' is the edge length of